LETTER TO THE SHAREHOLDERS
December 15, 2025
Napoleonville, LA IFUS:OTCID
In our last Shareholder Update we shared a customer testimonial that reported of the efficacy of IFUS Intact Digest™ with Nutri-Mastic™ applied orally and/or topically to an outbreak of Eczema.
In this update, our focus shifts to customer testimonials of the efficacy of IFUS Intact Digest™ with Nutri-Mastic™ applied orally and/or topically to an outbreak of Gout. We wish to acknowledge that this update is VERY science heavy, with an intent of offering what we are discovering through previously (mostly recently) published studies.
Here is but one example of an email remitted to IFUS from a customer suffering from Gout:
“Since I began taking Intact Digest in mid-February of 2025, I have had far less night time muscles cramps (“a.k.a. charlie horses”), in fact almost none except after I’ve done a 25-mile bike ride. I will often take another dose of Intact Digest if it has been a hot day and I’m dehydrated and feel I’m lacking electrolytes. I’ve also had NO episodes of gout which means I’m no longer afraid to eat pork, sausage and shrimp. Yay! I used to have gout attacks regularly. Those are the two main benefits I’ve noticed after starting Intact Digest although there are possibly others. I didn’t suffer a lot from indigestion prior but I’ve also had NO indigestion and good GI/bowel symptoms since beginning Intact Digest. One other symptom that I can’t explain is that I used to experience a lot of light-headedness after bending over, then standing up. That has disappeared for some reason. I’m not sure whether to give Intact Digest the credit for that and am going to wait a few more months to see if those symptoms recur. There is some medical evidence that I have found online that magnesium can play a role in stabilizing blood pressure. Whether it was taking Intact Digest or some other random reason, I’m certainly happy that is gone since it bothered me immensely putting the tee in playing golf and then having to wait for 15 seconds for the light-headedness to disappear.”
What is Gout?

Source: https://www.verywellhealth.com/gout-7105787
Per the Mayo Clinic: “Gout is a common and complex form of arthritis that can affect anyone. It’s characterized by sudden, severe attacks of pain, swelling, redness and tenderness in one or more joints, most often in the big toe.
An attack of gout can occur suddenly, often waking you up in the middle of the night with the sensation that your big toe is on fire. The affected joint is hot, swollen and so tender that even the weight of the bedsheet on it may seem intolerable.
Gout symptoms may come and go, but there are ways to manage symptoms and prevent flares.”
Source: https://www.mayoclinic.org/diseases-conditions/gout/symptomscauses/syc-20372897

Source: https://www.annaiarulhospital.com/causes-of-gout/
Our IFUS Scientific Team offers additional information about Gout:
What causes Gout?
The Cleveland Clinic tells us:
| A buildup of excess uric acid in your body causes gout. Your body naturally makes uric | |||||||||||||
| acid when it breaks down chemicals called purines found in certain foods and drinks. | |||||||||||||
| Your kidneys usually filter uric acid out of your blood, and then it leaves your body when | |||||||||||||
| you pee. Sometimes your body makes too much uric acid, or your kidneys don’t remove it from | |||||||||||||
| your blood fast enough. When your body has | high levels of uric acid (hyperuricemia) | , | |||||||||||
| uric acid crystals can build up and settle into your joints. The sharp crystals clump | |||||||||||||
| together and cause sudden episodes of pain, swelling and other symptoms. Having temporarily high uric acid levels doesn’t mean you’ll definitely develop gout. Many people with hyperuricemia never get gout. Gout risk factors Men Gout can affect anyone. are three times more likely to develop gout. Women | |||||||||||||
| usually don’t experience gout until after | menopause | . People with certain health | |||||||||||
| conditions are more likely to develop gout, including: | |||||||||||||
| • | Overweight or | obesity | . | ||||||||||
| ••••• | Congestive heart failure | .high blood pressure). | |||||||||||
| Diabetes | . | ||||||||||||
| Hypertension ( | |||||||||||||
| Kidney disease | . | ||||||||||||
| Blood cancer | . | ||||||||||||
You’re more likely to experience gout if you:
| • | Have a biological parent or grandparent who has gout. | |||||||||
| • | Eat a lot of animal proteins — especially animal flesh, shellfish and foods that contain organ meat. | |||||||||
| • | Drink alcohol regularly. | |||||||||
| •• | Take a diuretic medication (water pills). | |||||||||
| Take | immunosuppressants | . | ||||||||
| Which foods cause gout? | ||||||||||
| Eating or drinking foods high in purines are more likely to lead to high uric acid levels in your body that cause gout, including: | ||||||||||
| • | Sugary drinks and sweets: | Standard table sugar is half fructose (fruit sugar), | ||||||||
| which breaks down into uric acid. Any food or drink with high sugar content can trigger gout. | ||||||||||
| • | High fructose corn syrup: This is a concentrated form of fructose. Packaged food products and processed snacks can contain lots of high fructose corn syrup. | |||||||||
| • | Alcohol: Even though not all alcoholic drinks are high in purines, alcohol prevents your kidneys from eliminating uric acid, pulling it back into your body, where it continues to accumulate. | |||||||||
| • | Organ meats: These include liver, tripe, sweetbreads, brains and kidneys. | |||||||||
| • | Game meats: Specialties such as goose, veal and venison all contain high levels | |||||||||
| •••• | of purines. | |||||||||
| Certain seafood: | Herring, scallops, mussels, codfish, tuna, trout and haddock. Beef, lamb, pork and bacon. Especially processed deli turkey. | |||||||||
| Red meat: | ||||||||||
| Turkey | : | |||||||||
| Gravy and meat sauces. | ||||||||||
Source: https://my.clevelandclinic.org/health/diseases/4755-gout
A deeper dive into the science behind what causes Gout reveals the following:
“Abstract: Gout is the most common inflammatory arthritis in adults, associated with hyperuricemia and the chronic deposition of monosodium urate (MSU) crystals. Hyperuricemia results from increased production of uric acid and decreased excretion by the kidneys and intestines. Urate excretion is regulated by a group of urate transporters, and decreased renal or intestinal excretion is the primary mechanism of hyperuricemia in most people. Genetic variability in these urate transporters is strongly related to variances in serum urate levels. Not all individuals with hyperuricemia show deposition of MSU crystals or develop gout. The initiation of the inflammatory response to MSU crystals is mainly mediated by the nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing protein 3 (NLRP3) inflammasome. The activated NLRP3 inflammasome complex cleaves pro-interleukin-1β (IL-1β) into its active form, IL-1β, which is a key mediator of the inflammatory response in gout. IL-1β leads to the upregulation of cytokines and chemokines, resulting in the recruitment of neutrophils and other immune cells. Neutrophils recruited to the site of inflammation also play a role in resolving inflammation. Aggregated neutrophil extracellular traps (NETs) trap and degrade cytokines and chemokines through NET-bound proteases, promoting the resolution of inflammation. Advanced gout is characterized by tophi, chronic inflammatory responses, and structural joint damage. Tophi are chronic foreign body granulomalike structures containing collections of MSU crystals encased by inflammatory cells and connective tissue. Tophi are closely related to chronic inflammation and structural damage.” (See Figure 1 and Figure 2:)
Source: Ahn EY, So MW. The pathogenesis of gout. J Rheum Dis. 2025 Jan 1;32(1):8-16. doi: 10.4078/jrd.2024.0054. Epub 2024 Nov 6. PMID:
39712248; PMCID: PMC11659655.
Figure 1. J Rheum Dis. 2024 Nov 6;32(1):8–16. doi: 10.4078/jrd.2024.0054
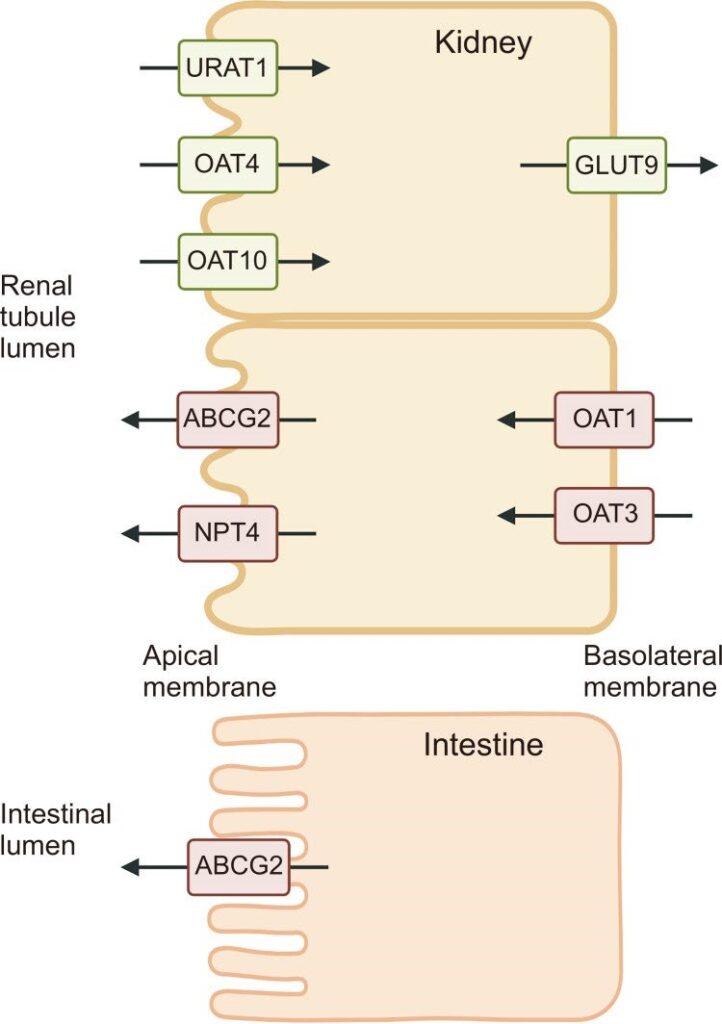
Urate transporters in kidneys and intestines. There are resorptive and secretory urate transporters. In the renal proximal tubule, URAT1, OAT4, OAT10 and GLUT9 act as resorptive urate transporters and ABCG2, NPT4, OAT1 and OAT3 function as secretory urate transporters. ABCG2 is also strongly associated with intestinal urate excretion. URAT1: urate transporter 1, OAT: organic anion transporter, GLUT9: glucose transporter 9, ABCG2: adenosine triphosphatebinding cassette super-family G member 2, NPT4: Na+-phosphate transporter 4.”
Figure 2. J Rheum Dis. 2024 Nov 6;32(1):8–16. doi: 10.4078/jrd.2024.0054
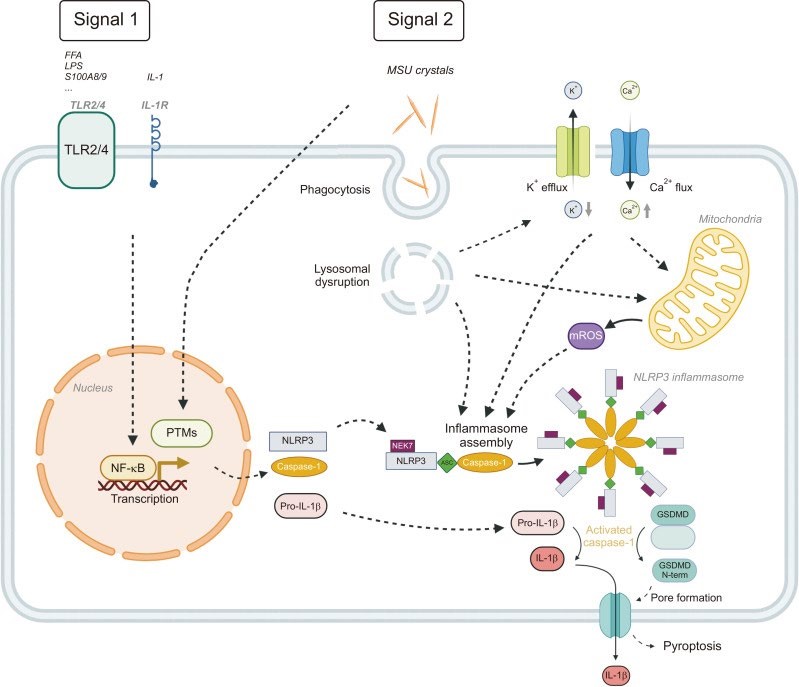
Priming and activation of the NLRP3 inflammasome in gout. Signal 1, the priming process, is mediated by TLRs (TLR2 or TLR4) or cytokine receptors through NF-kB activating pathways. This process controls the gene expression of pro-IL-1β and components of the NLRP3 inflammasome through upregulation of transcriptional level and PTM, preparing cells for inflammasome assembly. In Signal 2, phagocytosis of MSU crystals trigger the assembly of the NLRP3 inflammasome complex and activates caspase-1. Several mechanisms, including ionic K+ efflux, Ca2+ signaling, lysosomal disruption and mitochondrial reactive oxygen generation are known to involve in this process. Activated caspase-1 cleaves pro-IL-1β into IL-1β and also cleaves GSDMD into its amino-terminal fragment (GSDMD N-term) forms pores which facilitates IL-1β release and pyroptosis. TLRs: Toll-like receptors, NF-kB: nuclear factor-κB, IL-1β: interleukin-1β, NLRP3: nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing protein 3, MSU: monosodium urate, PTMs: post-translational modifications, mROS: mitochondrial reactive oxygen species, NEK7: NIMA-related kinase 7, ASC: apoptosisassociated speck-like protein, GSDMD: gasdermin D, NIMA: never in mitosis gene A. Revised from the article of Kingsbury et al. (J Inflamm Res 2011;4:39-49) [37], Bauernfeind et al. (J Immunol 2009;183:787-91) [42], Netea et al. (Blood 2009;113:2324-35) [43], Hornung et al. (Nat Immunol 2008;9:847-56) [51], So and Martinon (Nat Rev Rheumatol 2017;13:639-47) [52], and Kim (J Rheum Dis
Per your IFUS Scientific Team, a sample of key take aways from this information (where Gout is concerned) include:
- High levels of Uric Acid.
- Per Figure 1, the involvement of the kidneys and the intestine.
- Per Figure 2, “Priming and activation of the NLRP3 inflammasome in gout. Signal 1, the priming process, is mediated by TLRs (TLR2 or TLR4) or cytokine receptors through NF-kB activating pathways. This process controls the gene expression of pro-IL-1β and components of the NLRP3 inflammasome through upregulation of transcriptional level and PTM, preparing cells for inflammasome assembly.”
- MSU crystals is mainly mediated by the nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing protein 3 (NLRP3) inflammasome.
- K(+) Efflux / Ca(2+) Flux.
- The interrelationship between excess fructose and uric acid levels.
- And, per research presented later, the inter-relationship between Gout and IBF. Plus, Vitamin C supplementation could be considered as preventive measures, as those can lower serum uric acid levels as well as the risk of gout, per information on preventive measures for gout.
Can Gout be cured?
The Cleveland Clinic tells us that: “There’s no cure for gout. You’ll experience fewer attacks once you work with a healthcare provider to find treatments that manage your symptoms and lower your uric acid levels.”
Source: https://my.clevelandclinic.org/health/diseases/4755-gout
Can Gout be Managed?
Per a study performed by M. Kakutani-Hatayama, et.al., we find
“Nonpharmacological Management of Gout and Hyperuricemia: Hints for Better Lifestyle.”
Source: Kakutani-Hatayama M, Kadoya M, Okazaki H, Kurajoh M, Shoji T, Koyama H, Tsutsumi Z, Moriwaki Y, Namba M, Yamamoto T. Nonpharmacological Management of Gout and Hyperuricemia: Hints for Better Lifestyle. Am J Lifestyle Med. 2015 Sep 2;11(4):321-329. doi:
10.1177/1559827615601973. PMID: 30202351; PMCID: PMC6125106.
“Abstract
We reviewed lifestyle factors that influence serum uric acid levels and risk of gout flare, and how to improve their deleterious effects. Since obesity increases uric acid and weight gain increases gout risk, weight reduction by daily exercise and limiting intake of excess calories is recommended. However, strenuous exercise, which causes adenine nucleotide degradation; starvation, which decreases uric acid excretion; and dehydration may raise the level of uric acid in serum and trigger gout. Increased intake of purine-rich foods, such as meat and seafood, raise the level of uric acid in serum and is associated with increased risk of gout, whereas dairy products, especially low-fat types, are associated with a lower risk of gout. Also, heavy alcohol drinking raises the uric acid level and increases the risk of gout through adenine nucleotide degradation and lactate production. Sweet fruits and soft drinks containing fructose should be moderated, since fructose may raise uric acid and increase gout risk through uric acid production and/or decreased excretion. On the other hand, the Mediterranean diet is recommended for gout patients, since it may also help prevent hyperuricemia. Furthermore, coffee and vitamin C supplementation could be considered as preventive measures, as those can lower serum uric acid levels as well as the risk of gout.”
On every package, please note that IFUS recommends the use of Intact Digest™ in orange juice.
Medical Treatment of Gout:
“When you have gout, your doctor may recommend medications to manage the underlying cause of gout and treat active gout flares.
To treat a gout flare, your doctor may recommend medications including:
- Over-the-counter or prescription anti-inflammatory medications, which can reduce pain and swelling.
- Colchicine, which helps to relieve gout pain. This medication works best when taken when symptoms first appear.
Corticosteroids, which can help decrease inflammation, provide pain relief, and reduce joint swelling. Corticosteroids may be given by mouth or injected directly into the affected joint. Because they are potent medications, your doctor will prescribe the lowest dose possible to achieve the desired benefit.
Another key step to treating gout is managing hyperuricemia, which is the underlying cause of gout. There are medications available to lower urate levels, including:
- Xanthine oxidase inhibitors, which help prevent the production of urate.
- Uricosuric agents, which help the kidneys flush more urate out of the body.
- Uricase, which breaks down urate into a form that is easily removed from the body. This medication is recommended for people who have not responded to other medications.
Other treatments may include making diet and lifestyle changes to reduce risk factors you can control.”
Source: https://www.niams.nih.gov/health-topics/gout/diagnosis-treatmentand-steps-to-take
Is There Plausible Scientific Evidence that Supports the Efficacy of Intact Digest™ with Nutri-Mastic™ in the Relief of Gout?
In a study performed by O. Hamid, et.al, we find “Increased prevalence of gout in patients with inflammatory bowel disease: A population-based study.
Source: JGH Open. 2023 Sep 1;7(9):640-644. doi: 10.1002/jgh3.12963. PMID: 37744707; PMCID: PMC10517438.
“Abstract
Background and aim: Arthritis is a recognized extra-intestinal manifestation of inflammatory bowel disease (IBD). Studies show altered uric acid metabolism in IBD. This study aims to investigate the association between IBD and gout.
Methods: We used a multi-center database (Explorys Inc.) consisting of data from several US healthcare systems. We identified adults diagnosed with Crohn’s disease (CD) and ulcerative colitis (UC) between 1999 and 2022. In this cohort, we identified patients diagnosed with gout. We collected demographic data and identified patients diagnosed with IBD-associated arthritis and those who had intestinal resection. Risk factors associated with gout were collected. Multivariate analysis was used.
Results: Out of the 69 260 780 patients in the database, we identified 209 020 patients with UC (0.30%) of whom 9130 had gout (4.3%). Additionally, 249 480 had CD (0.36%) of whom 14 000 had gout (5.61%). Males were more prevalent in the UC and gout group than in the CD and gout group (58% vs 51%). After adjustment, CD was significantly associated with gout (odds ratio [OR] 1.68, confidence interval [CI]: 1.60-1.75). UC was also significantly associated with gout (OR 1.38, CI: 1.31-1.44). In subgroup analysis with intestinal resection, CD patients who had intestinal resection had higher association with gout versus those without surgery (OR 2.34, CI: 2.25-2.43). Similar increase was observed in the UC group with intestinal resection (OR 1.53, CI: 1.49-1.56).
Conclusion: IBD is strongly associated with gout, with higher correlation observed with CD. Intestinal resection is associated with an increase in the risk of gout. Patients with IBD who present with new-onset arthritis should be investigated for gout.”
Source: Hamid O, Alsabbagh Alchirazi K, Eltelbany A, Nanah R, Regueiro M. Increased prevalence of gout in patients with inflammatory bowel disease: A population-based study. JGH Open. 2023 Sep 1;7(9):640-644. doi: 10.1002/jgh3.12963. PMID: 37744707; PMCID: PMC10517438.
Hence, this study establishes a direct relationship between IBF and gout involving Uric Acid.
Additionally, a study performed by E. Papada, et.al., discusses the “Anti-oxidative
Efficacy of a Pistacia Lentiscus Supplement and Its Effect on the Plasma
Amino Acid Profile in Inflammatory Bowel Disease: A Randomised, DoubleBlind, Placebo-Controlled Trial.”
Source: Nutrients. 2018 Nov 16;10(11):1779. doi: 10.3390/nu10111779. PMID: 30453494; PMCID: PMC6267573.
“5. Conclusions: Given the increasing interest in the use of natural supplements with antioxidant and anti-inflammatory properties in IBD, herein the effects of a PL supplement on oxidative stress biomarkers in active IBD patients were explored. These results showed, for the first time, that PL could exhibit favourable effects in oxidative stress biomarkers in patients with active CD that could be of clinical importance.
Abstract: Oxidative stress is present in patients with Inflammatory Bowel Disease (IBD), and natural supplements with antioxidant properties have been investigated as a non-pharmacological approach. The objective of the present study was to assess the effects of a natural Pistacia lentiscus (PL) supplement on oxidative stress biomarkers and to characterise the plasma-free amino acid (AA) profiles of patients with active IBD (Crohn’s disease (CD) N = 40, ulcerative colitis (UC) N = 20). The activity was determined according to 5 ≤ Harvey Bradshaw Index ≤ 16 or 2 ≤ Partial Mayo Score ≤ 6. This is a randomised, double-blind, placebo-controlled clinical trial. IBD patients (N = 60) were randomly allocated to PL (2.8 g/day) or to placebo for 3 months being under no treatment (N = 21) or under stable medical treatment (mesalamine N = 24, azathioprine N = 14, and corticosteroids N = 23) that was either single medication (N = 22) or combined medication (N = 17). Plasma oxidised, low-density lipoprotein (oxLDL), total serum oxidisability, and serum uric acid were evaluated at baseline and follow-up. OxLDL/LDL and oxLDL/High-Density Lipoprotein (HDL) ratios were calculated. The plasma-free AA profile was determined by applying a gas chromatography/mass spectrometry analysis. oxLDL (p = 0.031), oxLDL/HDL (p = 0.020), and oxLDL/LDL (p = 0.005) decreased significantly in the intervention group. The mean change differed significantly in CD between groups for oxLDL/LDL (p = 0.01), and, in the total sample, both oxLDL/LDL (p = 0.015) and oxLDL/HDL (p = 0.044) differed significantly. Several changes were reported in AA levels. PL ameliorated a decrease in plasma-free AAs seen in patients with UC taking placebo. In conclusion, this intervention resulted in favourable changes in oxidative stress biomarkers in active IBD.”
Source: Papada E, Forbes A, Amerikanou C, Torović L, Kalogeropoulos N,
Tzavara C, Triantafillidis JK, Kaliora AC. Antioxidative Efficacy of a
Pistacia Lentiscus Supplement and Its Effect on the Plasma Amino Acid
Profile in Inflammatory Bowel Disease: A Randomised, Double-Blind, Placebo-Controlled Trial. Nutrients. 2018 Nov 16;10(11):1779. doi:
10.3390/nu10111779. PMID: 30453494; PMCID: PMC6267573.
Excerpts from the aforementioned study provide insight into PL (or Pistacia lentiscus):
- “Amino acids (AAs) control and protect intestinal health, and not only act as substrates for proteinosynthesis in intestinal mucosal cells [10], but also as regulators of major metabolic pathways [11,12]. Their protective function may be linked to intestinal epithelial cells apoptosis and proliferation, as well as inhibition of inflammation and oxidative stress through blocking of the NF-κB signaling pathway [13]. Recently, it has been shown that variations in the levels of some AAs may be involved in the pathogenesis of IBD [14]. Data on circulating AAs in active IBD patients are limited and sometimes conflicting; however, it seems that the AA profile differentiates IBD patients from healthy controls, pointing towards a link between the AAs and the disease process [15,16,17]. Aminograms and multivariate indexes using AA molar ratios have been proven to be useful in reflecting the nutritional status, assessing disease activity, and monitoring disease progression [15,18].”
- “Pistacia lentiscus L. var latifolius Coss or Pistacia lentiscus var. Chia (PL) is an evergreen shrub of the pistacia genus that is cultivated for its aromatic resin mainly on the Greek island of Chios. Previous studies of Pistacia lentiscus resin have documented the presence of phenolics [19], terpenes [20,21], their bioavailability in humans [22], as well as antioxidant [23], anti-inflammatory [24], lipid-lowering [25], and immunomodulating properties [26,27] of this natural product.”
- “The European Medicines Agency has recognised Pistacia lentiscus resin as a herbal medicinal product with the following indications: (a) mild dyspeptic disorders, (b) symptomatic treatment of minor inflammations of the skin, and (c) as an aid in the healing of minor wounds [28].”
- “In previous research, it has been reported that a 3-month intervention with a supplement containing the natural resinous product of Pistacia lentiscus (the supplement herein given as PL), adjunctive to stable medical treatment, regulated faecal biomarkers and improved quality of life in active IBD patients compared with a placebo [29]. Herein, the aim was to explore the effects of this intervention on biomarkers of oxidative stress in active IBD. Additionally, another study in healthy adults has shown that AA profiles are modified in response to acute supplementation with PL resin [30]. AAs are key regulators in metabolic pathways that affect intestinal health, and are also implicated in oxidative stress and inflammatory cytokines production [31,32]. Therefore, a second aim was to assess whether PL administration could have an impact on the health of patients reflected in the plasma-free AA profile after an assessment of AAs in the diet.”
Is there evidence of a specific biochemical pathway that supports the efficacy of Intact Digest™ with Nutri-Mastic™ in providing relief for those who suffer from Gout?
In Figure 2 above, we find the “Priming and activation of the NLRP3 inflammasome in gout. Signal 1, the priming process, is mediated by TLRs (TLR2 or TLR4) or cytokine receptors through NF-kB activating pathways. This process controls the gene expression of pro-IL-1β and components of the NLRP3 inflammasome through upregulation of transcriptional level and PTM, preparing cells for inflammasome assembly.”
Furthermore, in the study performed by Papada E, et.al, we find the
“Antioxidative Efficacy of a Pistacia Lentiscus Supplement and Its Effect on the Plasma Amino Acid Profile in Inflammatory Bowel Disease: A
Randomised, Double-Blind, Placebo-Controlled Trial.”
Source: Nutrients. 2018 Nov 16;10(11):1779. doi:
10.3390/nu10111779. PMID: 30453494; PMCID: PMC6267573.
As a reminder: “Amino acids (AAs) control and protect intestinal health, and not only act as substrates for proteinosynthesis in intestinal mucosal cells [10], but also as regulators of major metabolic pathways [11,12]. Their protective function may be linked to intestinal epithelial cells apoptosis and proliferation, as well as inhibition of inflammation and oxidative stress through blocking of the NF-κB signaling pathway [13]. Recently, it has been shown that variations in the levels of some AAs may be involved in the pathogenesis of IBD [14]”
The study also tells us, “Out of the 60 patients, 27 (45.0%) were randomised to the placebo group and 33 (55.0%) to the verum group, while 40 (66.7%) of them were diagnosed with CD and 20 (33.3%) with UC.
The decreased levels of oxLDL after PL intervention could point towards an antioxidant effect of this natural supplement in active IBD patients. Future research on its mechanism of action is justified, since its main bioactive compounds, which are mostly triterpenes (i.e., mastihadienonic and isomastihadeinonic acids), are bioavailable and exhibit antioxidant effects in vivo [22]
Another interesting result of the present study was the significant reduction in the ratios of oxLDL/LDL and oxLDL/HDL in the PL arm compared with the placebo arm. A decrease in feacal lysozyme in the PL arm has been reported in the study of Papada and coworkers [29], and this parallels the decrease in the oxLDL/LDL and oxLDL/HDL ratios.
However, some stimulating results regarding AAs changes in UC have been revealed. As such, levels of allo-isoleucine, isoleucine, cysteine, and tryptophane significantly decreased in the placebo group, whereas in the PL group they remained unchanged, with the mean changes between the groups being significant.
The fact that AA alterations were present predominantly in the group of UC patients may indicate a more favourable effect of PL in patients with active UC.”
Yet, another question asked by the IFUS Scientific Team involves how are leucinerich repeat- and pyrin domain-containing protein 3 (NLRP3) inflammasome regulated in the body.
The science disusses: “DNA methylation, histone alterations, and microRNAs. The regulation of the NLRP3 inflammasome involves a complex interplay of various factors, including DNA methylation, histone alterations, and microRNAs. These epigenetic mechanisms play a crucial role in modulating the activation and expression of the NLRP3 inflammasome, which is essential for understanding its function in inflammatory and neurodegenerative diseases.”
Source: kodi, T., Sankhe, R., Gopinathan, A. et al. New Insights on NLRP3 Inflammasome: Mechanisms of Activation, Inhibition, and Epigenetic Regulation. J Neuroimmune Pharmacol 19, 7 (2024).
https://doi.org/10.1007/s11481-024-10101-5
In a study conducted by S.Paik, et.al., “Updated insights into the molecular networks for NLRP3 inflammasome activation.”
Source: Paik S, Kim JK, Shin HJ, Park EJ, Kim IS, Jo EK. Updated insights into the molecular networks for NLRP3 inflammasome activation. Cell Mol Immunol. 2025 Jun;22(6):563-596. doi: 10.1038/s41423-025-01284-9. Epub 2025 Apr 30. PMID: 40307577; PMCID: PMC12125403.
We begin with Fig. 1 provided by the S.Paik team, noting that involvement of many elements like cytokines (in this case TNF specifically):
Fig. 1. Cell Mol Immunol. 2025 Apr 30;22(6):563–596. doi:
10.1038/s41423-025-01284-9
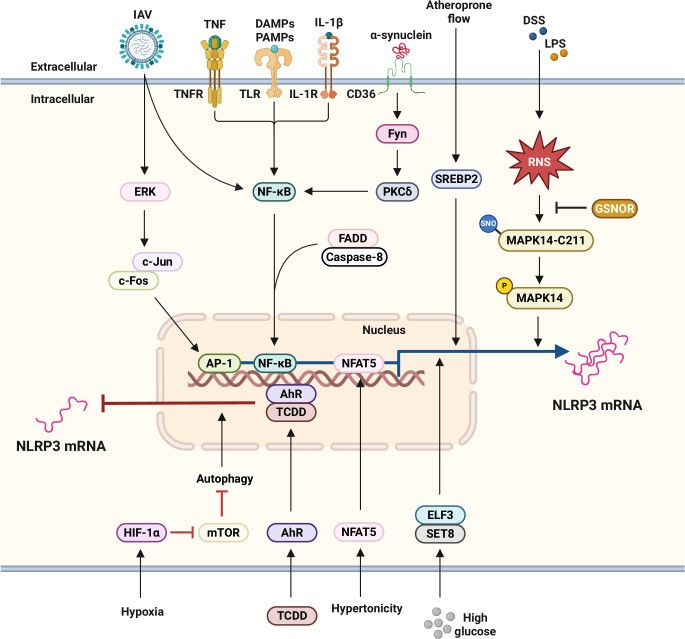
Transcriptional regulation of NLRP3 expression. The activation of TLRs by DAMPs, PAMPs, and pro-inflammatory cytokines such as TNF and IL-1β triggers NF-κB activation, which subsequently induces the transcription of the NLRP3 gene. This NF-κB-dependent transcription is mediated by FADD and caspase-8. During IAV infection, NLRP3 transcription is also upregulated via the ERK/c-Jun/AP-1 signaling pathway. In response to α-synuclein, Fyn kinase regulates PKCδ-mediated NF-κB activation, thereby promoting NLRP3 expression. Furthermore, atheroprone flow activates the NLRP3 inflammasome through SREBP2 activation. Under hypertonic stress, NFAT5 functions as a transcription factor to enhance NLRP3 expression. In endothelial cells exposed to high glucose conditions, ELF3 interacts with SET8 to regulate NLRP3 transcription. Conversely, during TCDD exposure, AhR suppresses NLRP3 expression. In models of LPS-induced septic shock and DSS-induced colitis,
GSNOR modulates NLRP3 expression by reducing S-nitrosylated MAPK14 levels. Under hypoxic conditions, the accumulation of HIF-1α inhibits NLRP3 transcription by downregulating mTOR signaling and promoting autophagy, particularly in the context of DSS-induced colitis. AhR aryl hydrocarbon receptor, DAMP damage-associated molecular pattern, DSS dextran sodium sulfate, ELF3 transcription factor E74-like factor 3, FADD fas-associated death domain,
GSNOR S-nitrosoglutathione reductase, HIF-1α hypoxia-inducible factor 1-alpha, IAV influenza A virus, LPS lipopolysaccharide, MAPK14 mitogen-activated protein kinase 14, mTOR mammalian target of rapamycin, NFAT5 nuclear factor of activated T cells 5, PAMP pattern-associated molecular pattern, PKCδ protein kinase Cδ, RNS reactive nitrogen species, SNO S-nitrosation, SREBP2 sterol regulatory element-binding protein2, TCDD 2,3,7,8-tetrachlorodibenzop-dioxin, TLR toll-like receptor, TNF tumor necrosis factor, TNFR tumor necrosis factor receptor.
The impact of Chios Mastic Gum is becoming more viably established by research like that performed by A. Triantafyllou A, et.al., Anti-inflammatory activity of Chios mastic gum is associated with inhibition of TNF-alpha induced oxidative stress.
(Nutr J. 2011 Jun 6;10:64. doi: 10.1186/1475-2891-10-64. PMID:
21645369; PMCID: PMC3127998.)
“Results
Spin trapping study did not show significant scavenging of superoxide by mastic gum itself. However, mastic gum inhibited cellular production of superoxide and H2O2 in dose dependent manner in TNF-α treated rat aortic smooth muscle cells but did not affect unstimulated cells. TNF-α significantly increased the cellular superoxide production by NADPH oxidase, while mastic gum completely abolished this stimulation. Mastic gum inhibited the activity of purified PKC, decreased PKC activity in cell homogenate, and attenuated superoxide production in cells stimulated with PKC activator PMA and PKC-dependent angiotensin II in endothelial cells.”
Source: Triantafyllou A, Bikineyeva A, Dikalova A, Nazarewicz R, Lerakis S, Dikalov S. Anti-inflammatory activity of Chios mastic gum is associated with inhibition of TNF-alpha induced oxidative stress. Nutr J. 2011 Jun 6;10:64. doi: 10.1186/1475-2891-10-64. PMID: 21645369; PMCID:
PMC3127998.
We continue. Fig. 2 provided by the S.Paik team reveals the involvement of many elements (in this case K(+) and Ca(2+) as well as the impact on mitochondria):
Fig. 2. Cell Mol Immunol. 2025 Apr 30;22(6):563–596. doi:
10.1038/s41423-025-01284-9

Second signal of NLRP3 inflammasome activation. A Perturbation of ion homeostasis perturbation. Membrane damage induced by Mycobacterium tuberculosis and TLR4 activation triggered by ornithine lead to K⁺ efflux. In addition, Ca²⁺ influx through the mechanosensitive channel PIEZO is converted into K⁺ efflux via KCNN4, thereby promoting NLRP3 inflammasome activation following stimulation with LPS and Yoda1. K⁺ efflux is further facilitated by various ion channels, including Kv1.3, KCa3.1, and TREK-1, ultimately contributing to NLRP3 activation. HBV activates the NLRP3 inflammasome through both K⁺ efflux and Na⁺ influx. Moreover, extracellular histones induce Ca²⁺ influx and recruit TWIK2 to the plasma membrane, resulting in K⁺ efflux through the TWIK2 channel. Extracellular ATP, released via pannexin-1 channels, binds to P2X7 receptors, leading to both Ca²⁺ influx and K⁺ efflux. Subsequently, K⁺ efflux promotes Cl⁻ efflux through CLIC1 and CLIC4, further contributing to NLRP3 inflammasome activation. Additionally, Ca²⁺ influx via TRPM2 induces mtROS production, providing an additional activation signal. VRAC activation, ATP release, and subsequent P2YR activation also participate in this process. Together, these ionic fluxes converge to promote NLRP3 inflammasome activation. B Perturbation of mitochondrial homeostasis. Extracellular mtDNA, SFTSV, forchlorfenuron, and aristolochic acid I have been shown to induce mitochondrial dysfunction. Gasdermin processing is essential for the release of mtDNA into the cytosol. Imiquimod inhibits mitochondrial complex I of the ETC and NQO2, leading to the production of mtROS. In contrast, PCr, generated through ETC activity, helps maintain intracellular ATP levels. ROS generated via FADDosome induction, along with ox-mtDNA produced through the TLR4– CMPK2 signaling axis, translocate to the cytosol through the mPTP and VDAC, where they trigger NLRP3 inflammasome activation. Furthermore, cardiolipin interacts with NLRP3 to promote inflammasome assembly. The recruitment of proIL-1α to mitochondrial cardiolipin impairs mitophagy and further enhances NLRP3 inflammasome activation during LPS stimulation. C Perturbation of lysosomal homeostasis. Various stimuli—including imatinib, masitinib, LL-37, LLME, LPC, Candida albicans, H-ferritin, nicotine, particulate matter and crystals, carbon nanotubes, and lecithinase from Clostridium perfringens—induce lysosomal damage. This damage leads to the release of cathepsins into the cytosol, which subsequently triggers K⁺ efflux and activates the NLRP3 inflammasome. ATP adenosine triphosphate, Casp8 caspase-8, CLIC chloride intracellular channels, CMPK2 cytidine monophosphate kinase 2, ER endoplasmic reticulum, ETC electron transport chain, FADD fas-associated death domain, GSDMD gasdermin D, GSDME gasdermin E, HBV hepatitis B virus, H-ferritin, heavy chain-ferritin, IP3R2 inositol 1,4,5-trisphosphate receptor type 2, KCNN4 potassium–calcium-activated channel subfamily N member 4, LLME Leu-Leu-Omethyl ester, LPC lysophosphatidylcholine, LPS lipopolysaccharide, mPTP mitochondrial permeability transition pore, mtDNA mitochondrial DNA, N- N terminal, NQO2 quinone oxidoreductase 2, ox-mtDNA oxidized-mitochondrial DNA, PCr phosphocreatine, ROS reactive oxygen species, RTK receptor tyrosine kinase, SFTSV severe fever with thrombocytopenia syndrome virus, TLR4 tolllike receptor 4, TRPM2 transient receptor potential melastatin 2, VDAC voltagedependent anion channel, VRAC volume-regulated anion channel
Although the S.Paik team’s study provides in depth and invaluable information, we skip ahead to Fig. 5, which reveals the cascading effect on humans when cellular disruption occurs (dis-ease), especially on various organs in the body.
Fig. 5. Cell Mol Immunol. 2025 Apr 30;22(6):563–596. doi:
10.1038/s41423-025-01284-9
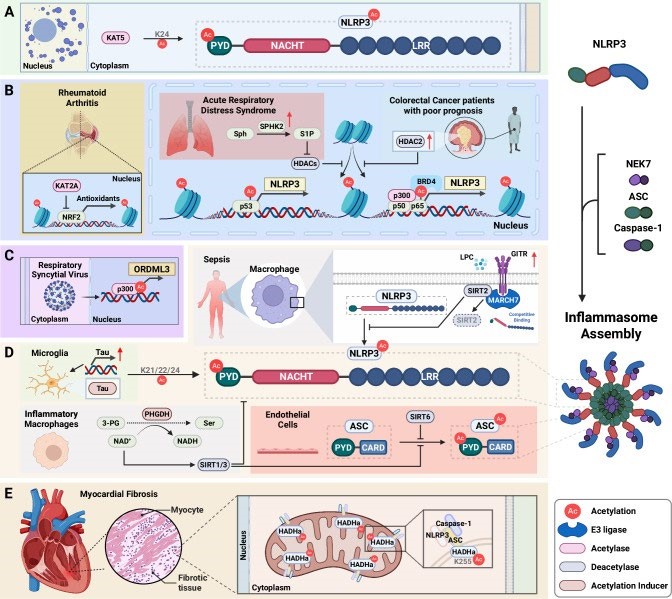
“Activation of NLRP3 inflammasome complex through acetylation or deacetylation. Posttranslational modifications via acetylation and deacetylation critically regulate NLRP3 inflammasome activity across various pathological conditions. A The acetyltransferase KAT5 directly acetylates NLRP3 at K24. B Rheumatoid arthritis, acute respiratory distress syndrome, and colorectal cancer. In rheumatoid arthritis, KAT2A promotes NLRP3 inflammasome activation by suppressing NRF2 transcriptional activity and downregulating antioxidant signaling. In acute respiratory distress syndrome, increased SPHK2 enhances S1P production, which in turn increases p53 acetylation, contributing to inflammasome activation. In colorectal cancer patients with poor prognosis, the upregulated deacetylase HDAC2 represses H3K27 acetylation, thereby modulating NLRP3 transcription through the BRD4/p-p65 complex. C Viral infections. During respiratory syncytial virus infection, histone hyperacetylation drives the upregulation of ORMDL3 expression, which in turn regulates NLRP3 expression levels. D Microglia in neurodegeneration, inflammatory cells, and endothelial cells. In microglia with elevated tau levels, tau protein acetylates the PYD domain of NLRP3 at K21, K22, and K24 residues. Additionally, PHGDH-mediated serine biosynthesis regulates the acetylation status of both NLRP3 and ASC via NAD+dependent modulation of SIRT1 and SIRT3 in inflammatory macrophages. In septic macrophages, upregulated GITR competitively binds to MARCH7 instead of NLRP3, leading to the degradation of the deacetylase SIRT2 and resulting in increased NLRP3 acetylation. In endothelial cells, SIRT6 suppresses ASC acetylation, thereby inhibiting inflammasome assembly. E Myocardial fibrosis. In myocardial fibrotic cells, acetylation of HADHa at K255 promotes NLRP3 inflammasome activation. ASC adaptor apoptosis-associated speck-like protein containing a caspase-recruitment domain, BRD4 Bromodomain-containing protein 4, GITR glucocorticoid-induced TNFR-related, HADHa hydroxyacyl-CoA dehydrogenase, HDACs histone deacetylases, HDAC2 histone deacetylase 2, KAT2A lysine acetyltransferase 2A, KAT5 lysine acetyltransferase 5, MARCH7 membrane-associated ring-CH-type finger 7, NRF2 nuclear factor erythroid 2related factor 2, ORMDL3 orosomucoid-like protein 3, PYD pyrin domain, S1P sphingosine-1-phosphate, SIRT2 sirtuin 2, SIRT6 sirtuin 6, SPHK2 sphingosine kinase 2.”
Hence, the IFUS Scientific Team once more begs additional questions as to the efficacy of Intact Digest™ with Nutri-Mastic™ on providing relief to individuals who suffer from Gout, IBF, or other similar related human ailments.
Is there an effect of phytochemicals (like those found in Chios Mastic Gum) on microRNAs.
In a study published by Hara Kang we find information on the, “MicroRNA-
Mediated Health-Promoting Effects of Phytochemicals” Source: Hara Kang, Int. J. Mol. Sci. 2019, 20(10), 2535; https://doi.org/10.3390/ijms20102535, Submission received: 29 April 2019 / Revised: 20 May 2019 / Accepted: 21 May 2019 / Published: 23 May 2019 “Abstract: Phytochemicals are known to benefit human health by modulating various cellular processes, including cell proliferation, apoptosis, and inflammation. Due to the potential use of phytochemicals as therapeutic agents against human diseases such as cancer, studies are ongoing to elucidate the molecular mechanisms by which phytochemicals affect cellular functions. It has recently been shown that phytochemicals may regulate the expression of microRNAs (miRNAs). MiRNAs are responsible for the fine-tuning of gene expression by controlling the expression of their target mRNAs in both normal and pathological cells. This review summarizes the recent findings regarding phytochemicals that modulate miRNA expression and promote human health by exerting anticancer, photoprotective, and anti-
hepatosteatosis effects. Identifying miRNAs modulated by phytochemicals and understanding the regulatory mechanisms mediated by their target mRNAs will facilitate the efforts to maximize the therapeutic benefits of phytochemicals.”

Additional science is found in work published by W. Chaiwangyen, “The Impact of Dietary Compounds in Functional Foods on MicroRNAs Expression.”
Source: Functional Foods – Phytochemicals and Health Promoting Potential. IntechOpen; 2021. http://dx.doi.org/10.5772/intechopen.96746
“Abstract: MicroRNAs (miRNAs) are a class of non-coding endogenous RNA molecules that are involved in post-transcriptional gene silencing via binding to their target messenger RNA, leading to mRNA degradation or translational repression. MicroRNAs can be modulated by several factors including hormones transcription factors, and dietary compounds. These biologically active compounds have positive impact on the progression of human pathology including noncommunicable diseases, which indicating that administration of diet may have potential as therapeutic agents in modulating the risk of chronic diseases. Interestingly, evidence emerging in recent years suggests that dietary miRNAs can be absorbed in human circulation, modulated human gene expression and biological functions. The exploitation of the miRNA functioning within different origins, cellular miRNAs and dietary miRNAs will help us to understand the molecular machinery as well as the regulatory mechanisms involved in fundamentally important biological processes. Therefore, this knowledge may be applied of natural bioactive compounds in preventive or therapeutic approaches.”
Summary:
Hence, what does the IFUS Scientific Team contend as to the efficacy of Intact Digest™ with Nutri-Mastic™ supplementation to those who suffer from Gout?
- High levels of Uric Acid possible reduced by Chios Mastic Gum o “Chios Mastic Gum (CMG) has been traditionally used for its antiinflammatory and antioxidant properties, which may help in managing conditions related to high uric acid levels.
- CMG is known for its potential to reduce symptoms of autoimmune diseases and has shown effects on lipid and glucose metabolism. o It has been used in traditional medicine for gastrointestinal disorders and has demonstrated therapeutic potential in various conditions. o Recent studies suggest that CMG may help in lowering plasma triglycerides and LDL cholesterol, which are factors associated with cardiovascular health.”
- While CMG shows promise in these areas, further research is needed to fully understand its effects on uric acid levels.
Ref. 1: Ottria, R.; Xynomilakis, O.; Casati, S.; Abbiati, E.; Maconi, G.; Ciuffreda, P. Chios Mastic Gum: Chemical Profile and Pharmacological Properties in Inflammatory Bowel
Disease: From the Past to the Future. Int. J. Mol. Sci. 2023, 24,
12038. https://doi.org/10.3390/ijms241512038
Ref. 2: Blomquist SA, Fernandez ML. Chios Mastic Gum: A Promising Phytotherapeutic for Cardiometabolic Health.
Nutrients. 2024 Sep 2;16(17):2941. doi: 10.3390/nu16172941. PMID: 39275256; PMCID: PMC11397435.
- Per Figure 1, the involvement of the kidneys and the intestines (and the liver), both of which have been shown to improve in various ways with the application of Chios Mastic Gum.
- “Chios Mastic Gum has been shown to have hepatoprotective effects and improve liver function. It has been found to support liver health and protect against metabolic liver damage. The hepatoprotective effects are considered safe at recommended doses and show promise in supporting the liver micro-environment and metabolic profile.
(Source: Masticlife) o Chios Mastic Gum also exhibits anti-inflammatory and antioxidative properties, which contribute to its beneficial effects on liver health. It has been found to reduce liver inflammation and support overall liver function. (Source: Advanced Orthomolecular Research)
- While Chios Mastic Gum has shown positive effects on liver health, it is important to note that some studies have indicated potential risks, such as harm to kidney and liver health. Therefore, it is advisable to consult a healthcare provider before using Chios Mastic Gum, especially under all circumstances. (Source Welzo)”
- Ref. 1: https://masticlife.com/en-us/pages/studieshepatoprotective-effects-of-chios-mastic
- Ref. 2: Soulaidopoulos, S.; Tsiogka, A.; Chrysohoou, C.;
- Ref. 1: https://masticlife.com/en-us/pages/studieshepatoprotective-effects-of-chios-mastic
Lazarou, E.; Aznaouridis, K.; Doundoulakis, I.; Tyrovola, D.; Tousoulis, D.; Tsioufis, K.; Vlachopoulos, C.; et al. Overview of Chios Mastic Gum (Pistacia lentiscus) Effects on Human Health. Nutrients 2022, 14, 590.
https://doi.org/10.3390/nu14030590
- Ref. 3: Soulaidopoulos, S.; Tsiogka, A.; Chrysohoou, C.;
Lazarou, E.; Aznaouridis, K.; Doundoulakis, I.; Tyrovola, D.; Tousoulis, D.; Tsioufis, K.; Vlachopoulos, C.; et al. Overview of Chios Mastic Gum (Pistacia lentiscus) Effects on Human Health. Nutrients 2022, 14, 590. https://doi.org/10.3390/nu14030590
- Per Figure 2, “Priming and activation of the NLRP3 inflammasome in gout. Signal 1, the priming process, is mediated by TLRs (TLR2 or TLR4) or cytokine receptors through NF-kB activating pathways. This process controls the gene expression of pro-IL-1β and components of the NLRP3 inflammasome through upregulation of transcriptional level and PTM, preparing cells for inflammasome assembly.” o This process affected by Chios Mastic Gum, albeit much research remains to be completed.
- MSU crystals is mainly mediated by the nucleotide-binding oligomerization domain-, leucine-rich repeat- and pyrin domain-containing protein 3 (NLRP3) inflammasome. Again, Chios Mastic Gum shown to affect the process in a positive way, albeit much research remains to be completed. o The impact of Chios Mastic Gum is becoming more viably established by research like that performed by A. Triantafyllou, et.al., Antiinflammatory activity of Chios mastic gum is associated with inhibition of TNF-alpha induced oxidative stress. (Note: TNF-alpha is seemingly interconnected to NLRP3 and other agents that create Gout and other diseases.)
Ref. 1: A. Triantafyllou, et.al., , Nutr J. 2011 Jun 6;10:64. doi:
10.1186/1475-2891-10-64. PMID: 21645369; PMCID: PMC3127998.)
- K+ Efflux / Ca+ Flux remains to be explored.
- “Vitamin C supplementation could be considered as preventive measures, as those can lower serum uric acid levels as well as the risk of gout, per information on preventive measures for gout.” o IFUS suggests that Nutri-Mastic be taken with Orange Juice as there is evidence that OJ has a positive effect on the efficacy of NutriMastic™. This begs a deeper question as to whether another scientific overlay is occurring here.
- Furthermore, “coffee and vitamin C supplementation could be considered as preventive measures, as those can lower serum uric acid levels as well as the risk of gout.”
Ref. 1: Source: Kakutani-Hatayama M, Kadoya M, Okazaki H, Kurajoh M, Shoji T, Koyama H, Tsutsumi Z, Moriwaki Y,
Namba M, Yamamoto T. Nonpharmacological Management of Gout and Hyperuricemia: Hints for Better Lifestyle. Am J Lifestyle Med. 2015 Sep 2;11(4):321-329. doi:
10.1177/1559827615601973. PMID: 30202351; PMCID:
PMC6125106.
- And, the inter-relationship between Gout and IBF, per a study performed by O. Hamid, et.al, we find “Increased prevalence of gout in patients with inflammatory bowel disease: A population-based study.
- Gout and IBF shared similar biomarkers.
- Chios Mastic Gum shown to impact IBF. “The fact that AA alterations were present predominantly in the group of UC patients may indicate a more favourable effect of PL in patients with active
UC.”
- Ref. 1: O. Hamid, et.al., JGH Open. 2023 Sep 1;7(9):640-644.
doi: 10.1002/jgh3.12963. PMID: 37744707; PMCID:
PMC10517438.
- Ref. 2: Papada E, et.al, “Antioxidative Efficacy of a Pistacia
Lentiscus Supplement and Its Effect on the Plasma Amino Acid
Profile in Inflammatory Bowel Disease: A Randomised,
Double-Blind, Placebo-Controlled Trial.” Source: Nutrients. 2018 Nov 16;10(11):1779. doi: 10.3390/nu10111779. PMID: 30453494; PMCID:
PMC6267573.
Is all of this information (science) pure coincidence, OR is it indicative of the plausible of efficacy of Intact Digest™ with Nutri-Mastic™?
The complexity of the science for which we are tasking our IFUS Scientific Team is expressed in the diagram below. Each dot represents a Metabolic Cycle. The lines represent simplified interconnections. Hence, the information presented in this update attempts to bring some level of clarity to only a couple of the dots and their respective interconnectivity.
Yet, while there is much that remains unknown, our IFUS Scientific Team is finding science, which at the metabolic level, provides plausible explanations that could explain why Intact Digest™ with Nutri-Mastic™ is being reported by customers to ease their symptoms. This is observational science at best and requires statistical patient trials to offer tangible proof. However, that there exists a possibility that our IFUS Product Lines (when applied as the technology that we believe each to be) demands that we continue our pursuit of scientific truth.
Please note Figure 17-A below:

Figure 17.1 A schematic of cellular metabolic pathways. As we learn about metabolism, we soon see that there are many common themes to cellular chemical transformations, whether we are considering processes as diverse as amino acid biosynthesis, steroid hormone metabolism, or DNA repair.
Source:https://ecampusontario.pressbooks.pub/bioc2580/chapter/introductio n-to-metabolism-metabolic-pathways/
This begs one last question in this line of inquiry, that being: Does Intact Digest™ with Nutri-Mastic™ contain plausible efficacy in the relief of Gout?
One could assert that without statistical patient trials the feedback from customers coupled with the science offered is nothing more than coincidence. In isolation, one could agree with this assertion. However, when considering the myriad of successes being reported from each of the IFUS Product Lines…all of which begin with Nutri-Mastic™…the reported successes warrant at a minimum consideration by the scientific community and beyond that Nutri-Mastic™ as a proprietary formulation and technology presents qualitative evidence similar to statistical trials being performed by researchers across the globe.
This contention is supported by the M. Shabani Team (“The relationship between gut microbiome and human diseases: mechanisms, predisposing factors and potential intervention,” Front Cell Infect Microbiol. 2025 May 6;15:1516010. doi: 10.3389/fcimb.2025.1516010.), the S. Soulaidopoulos and team provide similar information on “Therapeutic potentials of Chios Mastic.”
Figure 1. Therapeutic potentials of Chios Mastic.
Nutrients. 2022 Jan 28;14(3):590. doi: 10.3390/nu14030590
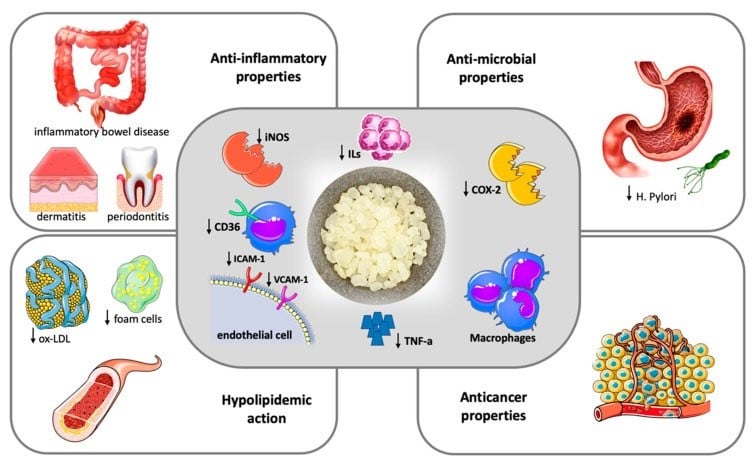
Per the diagram above, one could ask and summarize:
- Is “iNOS” involved with gout? “In summary, MSU induces iNOS expression that is associated with increased expression of inflammatory cytokines, oxidative stress, and edema. ERK1/2 or/and p38 may mediates MSUinduced iNOS expression. Suppression of iNOS could be a new therapeutic target for gout.”
- Ju TJ, et.al., Inhibition of Inducible Nitric Oxide Synthase Attenuates Monosodium Urate-induced Inflammation in Mice. Korean J Physiol Pharmacol. 2011 Dec;15(6):363-9. doi: 10.4196/kjpp.2011.15.6.363. Epub 2011 Dec 27. PMID: 22359474; PMCID: PMC3282224.
- Is “ICAM 1” involved with gout? “Conclusion: The MSU induced ICAM-1 expression on HRMCs and cell-cell adhesion involved TLR2/4-p38-ICAM1 pathway and TLR2/4 independent ASC-p38-ICAM1 axis. These findings might partly explain the mechanisms underlying gouty nephropathy.”
- Luo SF, et.al., Monosodium urate crystals induced ICAM-1 expression and cell-cell adhesion in renal mesangial cells:
Implications for the pathogenesis of gouty nephropathy. J Microbiol Immunol Infect. 2020 Feb;53(1):23-32. doi:
10.1016/j.jmii.2017.12.004. Epub 2018 Jan 31. PMID: 29657028.
- Is VCAM1 involved in gout? Fig. 5 below provides insight, whereby the
“leucocyte” is affected. (See “B”)
FIG. 5. Antioxid Redox Signal. 2011 Sep 15;15(6):1607–1638. doi: 10.1089/ars.2010.3522
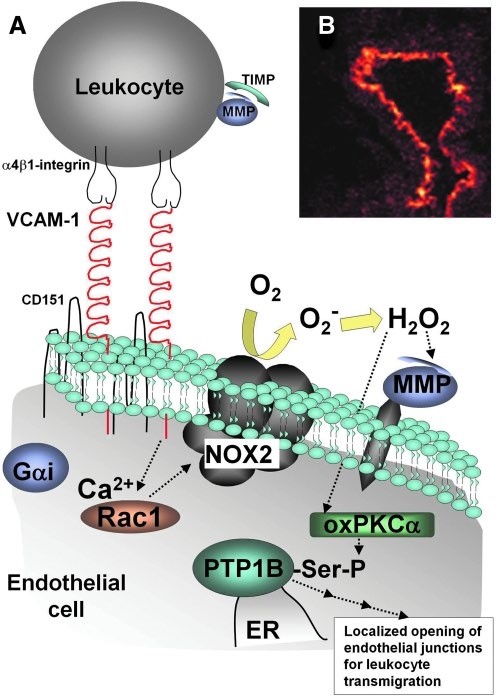
VCAM-1 signal transduction. (A) Model for VCAM-1 signaling. Crosslinking of
VCAM-1 activates calcium fluxes and Rac-1, which then activates endothelial cell
NOX2. Nox2 catalyzes the production of superoxide that then dismutates to H2O2. VCAM-1 induces the production of only 1 μM H2O2. H2O2 activates endothelial cell-associated MMPs that degrade extracellular matrix and endothelial cell surface receptors in cell junctions. The endothelial cell-derived
H2O2 also mediates a 2–5 h delayed activation of lymphocyte-associated MMPs by inducing the degradation of leukocyte TIMPs. H2O2 diffuses through membranes at 100 μm/s to activate p38MAPK. H2O2 also oxidizes and transiently activates endothelial cell PKCα. PKCα phosphorylates and activates PTP1B on the endoplasmic reticulum. PTP1B is not oxidized. These signals through ROS, MMPs, PKCα, and PTP1B are required for VCAM-1dependent leukocyte transendothelial migration. The G protein Gαi is also involved in VCAM-1 signaling. (B) Mouse lung tissue section from antigenchallenged lungs was labeled with anti-VCAM-1 and a TRITC-conjugated secondary antibody. VCAM-1 labels the luminal and lateral, but not the basal surface of vascular endothelial cells in vivo. ER, endoplasmic reticulum; H2O2, hydrogen peroxide; MMP, matrix metalloproteinase; oxPKCα, oxidized protein kinase Cα; PTP1B, protein tyrosine phosphatase 1B; ROS, reactive oxygen species; TIMP, tissue inhibitor of metalloproteinase.”
Once more, we find science that warrants further consideration as to the efficacy of Intact Digest™ with Nutri-Mastic™. It begs yet another question: Does Chios Mastic Gum affect VCAM-1 signal transduction?
“Chios Mastic Gum (CMG) has been shown to significantly decrease the expression of VCAM-1 and ICAM-1 in Human Aortic Endothelial cells (HAECs) when incubated with CMGNF and tirucallol. These substances, which are components of CMG, exert anti-inflammatory effects and inhibit the expression of adhesion molecules, which are crucial in the process of endothelial activation that is recognized as the primary event of the atherosclerotic process. This suggests that CMG may have a positive impact on VCAM-1 signal transduction, potentially influencing the development of cardiovascular diseases.” (See Figure 1.
Therapeutic potentials of Chios Mastic. Nutrients. 2022 Jan 28;14(3):590. doi:
10.3390/nu14030590)
Source: S. Soulaidopoulos, et.al., “Therapeutic potentials of Chios Mastic.” Front Cell Infect Microbiol. 2025 May 6;15:1516010.
doi: 10.3389/fcimb.2025.1516010.)

Source: Blomquist, S.A.; Fernandez, M.L. Chios Mastic Gum: A Promising Phytotherapeutic for Cardiometabolic Health. Nutrients 2024, 16, 2941.
“When I review diagrams from scientific studies like that which is presented immediately above, I challenge our IFUS Scientific Team to find evidence as to the efficacy and limitations of our IFUS Product Lines. Scientific truth can be a difficult task as indicated by the ‘MAP OF METABOLIC PATHWAYS’ presented above. However, we MUST ask the hard questions of ourselves so as to communicate with customers, who are being responsible and asking of us those very questions. We believe this creates a ‘market distinction’ that separates us from the competition. And, we are finding that as we use these ‘truths’ in both our marketing and sales efforts, we are realizing success. We are struck by evolving science, some of which we discovered as the update was being written. We are struck by the science, especially where Chios Mastic Gum is concerned. As a key component of our IFUS Product Lines, this natural ingredient with centuries of reported efficacy is used to formulate our proprietary product line and technology Nutri-Mastic™. When combined with our mineral formulation that we naturally source, our “secret sauce” so to speak becomes the basis of Nutri-Mastic™, as a proprietary formulation and technology, warranting consideration by scientific entities across the globe. Customer testimonials, qualitative and quantitative science we and our research partners are conducting, and any of scientific studies with patient trials on the ingredients found in Nutri-Mastic™ all seem to be far more than mere coincidence. Hence, the search for truth continues,” said Marc Walther, CEO of Impact Fusion International.
Once more, we are “Back to work!”
The information provided is for informational purposes only and is not intended as medical advice. Our products are not intended to diagnose, treat, cure, or prevent any medical condition. Always consult with a qualified healthcare professional before starting any new supplement, diet, or health regimen.
For our customers of both Intact Digest™ and Intact Endurance™ you may now send your testimonials to:
mwalther@impactfusionintl.com We can also be reached at 1-800-775-4130 seven days a week.
About Impact Fusion Internaonal Inc.
Impact Fusion Internaonal, Inc. is in the business of markeng products in the “Health and Wellness” sector of all internaonal markets. It is the company’s mission to invent, develop and market these proprietary products worldwide for the health and well-being of humans and animals.
The informaon contained in this release includes some statement that are not purely historical and that are “forward-looking statements.” Such forward-looking statements include, but are not limited to, statements regarding our and their management’s expectaons, hopes, beliefs, intenons or strategies regarding the future, including our financial condion, results of operaons. In addion, any statements that refer to projecons, forecasts or other characterizaons of future events or circumstances, including any underlying assumpons, are forward-looking statements. The words “ancipates,” “believes,”
“connue,” “could,” “esmates,” “expects,” “intends,” “may,” “might,” “plans,” “possible,” “potenal,” “predicts,” “projects,” “seeks,” “should,” “would” and similar expressions, or the negaves of such terms, may idenfy forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking. The forward-looking statements contained in this release are based on current expectaons and beliefs concerning future developments and the potenal effects on the pares and the corporate and administrave transacons. Forward-looking statements involve known and unknown risks, uncertaines and other factors, which may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied by the forward-looking statements and represent our management’s beliefs and assumpons only as of the date hereof. Except as required by law, we assume no obligaon to update these forward-looking statements, even if new informaon becomes available in the future.
Contact:
Impact Fusion Internaonal Inc.
204 Highway 1011
Napoleonville LA 70390
1-800-775-4130
Email: mwalther@impacusionintl.comhtps://www.impacusionbrands.com/brands
Updates can be found at the official Impact Fusion Twiter account @impacusionI
#Foodintelligence #NewMexico #healthiercatle #Screwworms
#Intact #Digeson #Endurance #Germany #Colorado
#legislaon #bagasse #drought #SUAREC #Louisiana
#greenhousegases #methanegas #catle #dairy #Texasfloods
#Texaswildfires $Waygu #India #Black Farmers Naonal
Associaon #Supreme AG™ #SGP+™ #Oklahoma
#KECO 96.5 FM radio #India #Australia #Brazil #Argenna #Canada #Vietnam

 My Account
My Account
You must be logged in to post a comment.